
Powerful, Easy-to-Use, Lightning-Fast
Supplier Quality Management Software
Connect with your suppliers and gain real-time visibility into inspection data across the supply chain. Speed-up new product introduction by streamlining FAI and PPAP processes. Simplify ongoing supply chain quality control with seamless data sharing and SCAR management.
Ensure quality at source, dramatically reduce the cost of poor quality, and save thousands of man-hours each year.
Customer Testimonials
The customer's perspective
“1factory has enabled a different level of integration with our key suppliers on high risk parts. We are solving the problem of stale and dusty control plans, and lack of supplier monitoring of quality data. We also found issues with measurement resolution on key features, and we found other cases where the distribution of data suggests abnormal processing.”
Semiconductor Capital Equipment Manufacturer
The supplier's perspective
“With 1factory, we give the customer access to the heart of RIVAL A/S. Everything is available and that gives the customer some security. The system saves us a lot of paperwork because we no longer have to make measurement reports and forward to the customer. We can also keep an eye on unmanned production on the weekends.”
Rival A/S, Denmark
Features: Supplier Quality Management
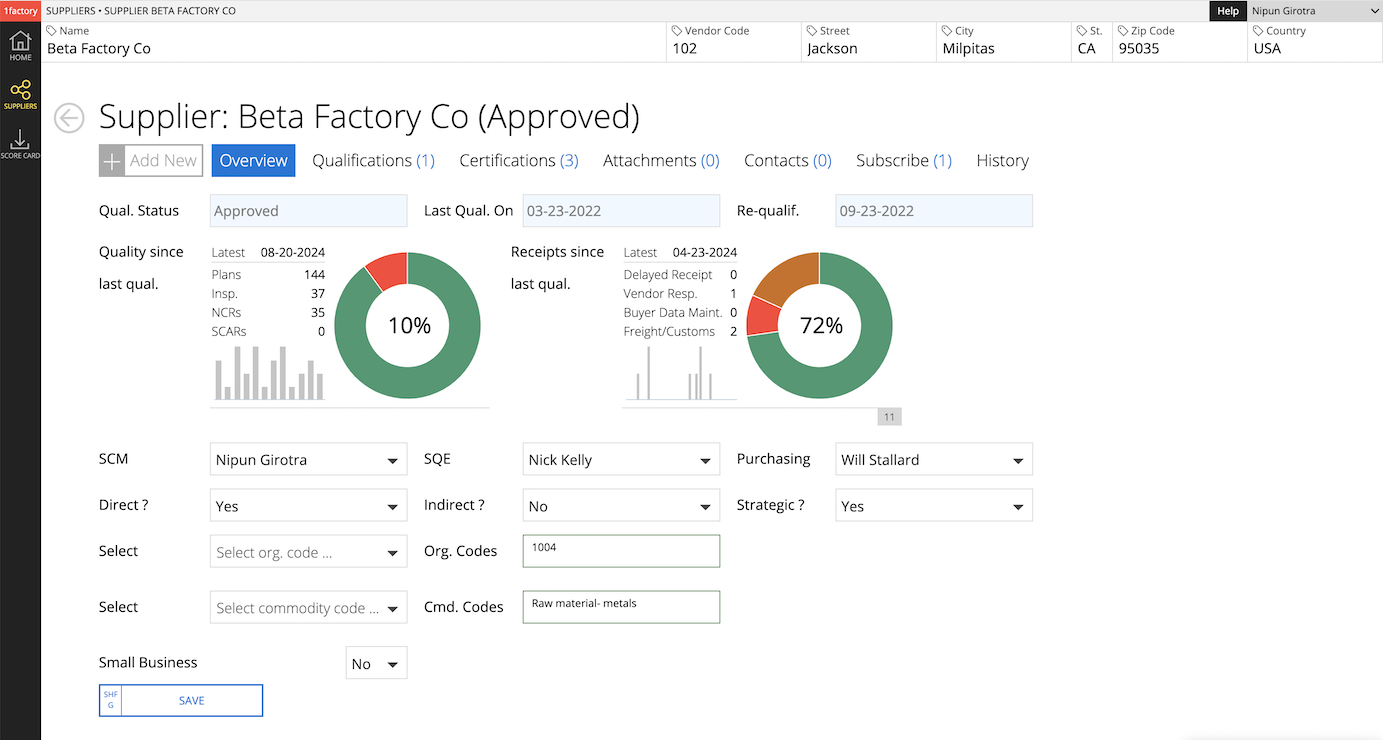
1 Approved Vendor List (AVL)
Simplify supplier evaluation and qualification. Manage supplier lifecycle from Onboarding through Approval. Monitor supplier quality and delivery performance.
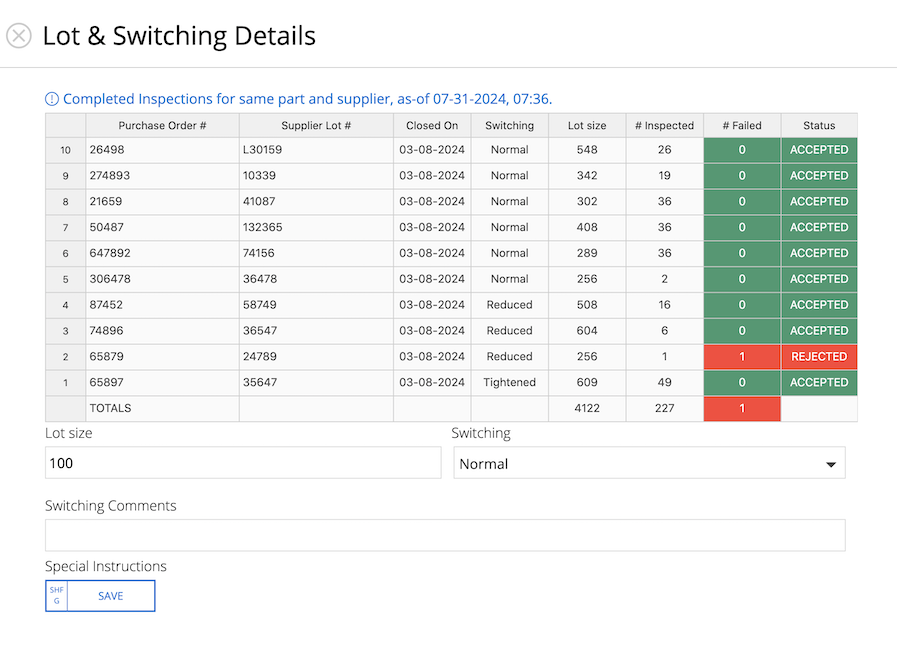
2 Incoming Inspections (IQC)
Inspect incoming materials. Capture visual and dimensional checks. Attach raw material & special processing certs. Use built-in lot switching rules.
3 Supplier Portal
Connect with your suppliers over the 1factory portal. Simplify FAI, PPAP, SCAR, and ongoing Supplier Quality Management. Collaborate to speed-up NPI.
4 Deep Supplier Collaboration
Connect with your suppliers and view their control plans and manufacturing inspection data in real-time to resolve problems before parts are shipped.
5 Supplier PPAP Management
Create and assign PPAPs to suppliers. Specify Levels, Requirements, and Due Dates. Review supplier PPAP documentation and approve individual elements.
6 Supplier Control Plans
Easily create and share quality control plans with suppliers. Ensure adequate controls for critical product features and process parameters.
7 Supplier First Articles
Speed-up the FAI process for suppliers with automated drawing ballooning and FAI report creation. Review and approve supplier FAIs.
8 Supplier Inspections
Verify measurements against specifications at the point-of-manufacturing. Assess risk before parts leave the supplier. Review and approve or reject inspections.
9 Supplier Corrective Actions
Request SCARs using the 8D process through the portal. Ensure all sections are filled-out and supplier actions are completed prior to closing.
10 Data: Customer to Supplier
Balloon drawings and assign control plans to your suppliers. Share incoming inspection data to ensure alignment where the OEM defines requirements.
11 Data: Supplier to Customer
Suppliers share plans, first articles, and real-time manufacturing inspection data. Ideal for situations where the supplier defines measurement frequency.
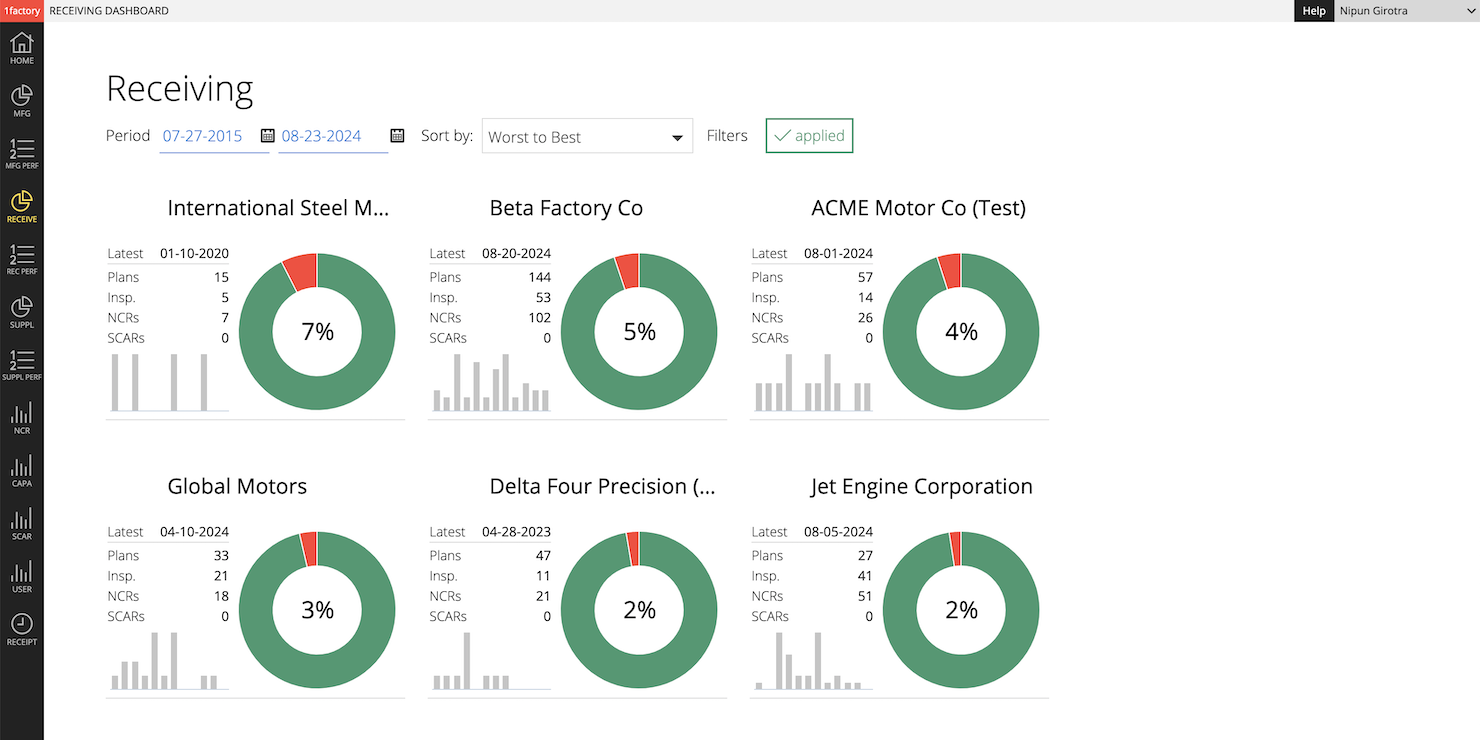
12 Dashboards & Scorecards
Manage performance in DPPM or Percentage terms. View feature-level SPC data (run-charts and histograms) to identify systemic issues.
Case Studies
Read Highlights & Transcript
Highlights: Source Quality & Global Collaboration
- Deep Supply Chain Integration: Zipline uses 1factory's Supplier Quality Management Software to connect with global suppliers, ensuring high-quality parts for life-saving drones.
- Portability & Speed: Standardizing Supplier FAI and Supplier PPAP submissions via the Supplier Portal has eliminated manual transcribing.
- Strategic Monitoring: By viewing supplier manufacturing data in real-time, Zipline identifies process anomalies (scrap, rework) before parts ship.
- User Experience: Suppliers were trained in under 2 hours, achieving immediate integration with critical supply chain tiers.
Transcript Excerpt: "1factory is the best SaaS software that Zipline uses."
Zipline - Autonomous Aircraft
Zipline designs, manufactures and operates the world's largest autonomous aircraft network, delivering life-saving supplies such as blood and medicines, and cutting delivery time from hours to minutes.
High-quality parts are critical to successful and reliable delivery. To ensure the quality of parts coming in from suppliers, Zipline deployed the 1factory system across its supply chain.
Today suppliers create Control Plans and First Articles and submit them to Zipline for review and approval via 1factory. Once Plans and FAIs are reviewed, suppliers submit on-going inspection data to Zipline via 1factory.
Watch a 7-minute video to hear first-hand from the Zipline team (Incoming Inspection, Quality Engineering, Supplier Quality, and Chief Technology Officer) about the power of 1factory, its ease-of-use, and its impact on cost of quality.
Easy to use. Lightning fast. Secure & reliable.
Intuitive design. Lightning-fast speed.
1factory's intuitive interface and streamlined workflows make training effortless - most teams achieve full deployment in under two hours with zero operational disruption.
Every interaction - from data entry to ballooning to search - happens instantaneously. Your manufacturing operations run at full pace, never waiting on software.
Secure. Reliable.
1factory meets the security standards that matter: ITAR-compliant hosting on AWS GovCloud, full NIST 800-171 compliance, and SOC 2 Type 2 certification.
Over the past decade, 1factory has maintained >99.99% uptime. Data encryption and daily database snapshots ensure your critical quality records are always protected.
FAQ: Supplier Quality Management
What challenges do manufacturers face in Supplier Quality Management?
Increasing product complexity and supply chain globalization make supplier quality control especially difficult. Most buyers lack real-time visibility into supplier production data, forcing quality teams to operate reactively rather than preventively. Challenges include:
Supplier quality processes are inconsistent. Without standardized processes across the supply chain, quality practices vary widely between suppliers and tiers, making it difficult to enforce requirements or compare performance.
Visibility into supplier production is limited. Buyers typically don't see quality data until parts arrive — or until defects surface in production — leaving little opportunity to intervene before problems escalate.
Traditional methods weren't built for global supply chains. Manual witness inspections and emailed spreadsheets were designed before global cloud networks existed, creating delays, gaps, and errors in quality data exchange.
New product introduction is slow and high-risk. Coordinating FAIs, PPAPs, and supplier approvals across multiple tiers is time-consuming and error-prone, delaying launches and increasing the risk of defects reaching production.
Supplier qualification and management lacks structure. Without a centralized system to qualify, monitor, and manage suppliers, approved vendor lists become outdated and supplier risk goes untracked.
Collecting supplier certifications is time-consuming and error-prone. For every incoming shipment, quality teams must track down and verify the correct certifications — raw material certs, outside processing certs, testing certs, and more. Without a systematic process, this becomes a manual scramble that consumes hours per shipment. For medical device manufacturers, missing or mismatched certs create gaps in the Device History Record that can halt production or trigger audit findings. For aerospace manufacturers, the stakes are equally high, as lot-level traceability is a fundamental compliance requirement.
The Impact
Product defects originating in the supply chain lead to production disruptions, wasted capacity, and costly escapes. Quality teams spend their time chasing down data and reacting to problems rather than preventing them, while the complexity of global supply chains continues to grow.
How does 1factory address Supplier Quality Management challenges? What is the Return on Investment?
1factory standardizes quality management processes across all suppliers regardless of tier, transforming reactive supplier management into proactive risk prevention through real-time visibility and automated data collection.
Standardized Supplier Processes: 1factory enforces consistent quality processes across every supplier and tier, ensuring all parties work to the same requirements, documentation standards, and inspection protocols.
Real-Time Production Visibility: The platform provides buyers with live access to supplier production data, enabling quality teams to monitor performance and identify issues before parts ever reach the factory floor.
Automated Data Collection: 1factory integrates directly with CMMs and Vision Systems to automate inspection data collection at the supplier, eliminating manual transcription and emailed spreadsheets.
Early Warning and Defect Prevention: A built-in early warning system uses defect risk analysis to flag potential escapes before shipment, shifting supplier quality from reactive containment to proactive prevention.
Supplier Collaboration for NPI and Production: 1factory enables seamless collaboration around control plans, FAIs, PPAPs, inspections, and SCARs — speeding up new product introduction and keeping actual inspection data, test results, and certifications accessible to buyers throughout ongoing production.
Approved Vendor List (AVL) Management: 1factory provides a centralized system to qualify, approve, and manage suppliers, ensuring the AVL stays current and supplier risk is continuously monitored.
Lot-Level Certificate Collection: For heavily regulated industries such as medical devices and aerospace, 1factory makes it easy to collect and manage supplier certifications for each lot, ensuring full traceability and audit readiness.
Return on Investment:
By replacing manual processes and disconnected spreadsheets with 1factory's Supplier Quality Management software, manufacturers can reduce defect escapes, accelerate new product launches, and gain the supply chain visibility needed to prevent production disruptions — without adding headcount or relying on reactive containment.
500+ Customers. 30+ Countries. 20+ Industries

AEROSPACE & INDUSTRIAL

AEROSPACE MACHINING

PRECISION MACHINING

MEDICAL DEVICES

AUTONOMOUS AIRCRAFT

ADVANCED CERAMICS